- Titan Medical (TMD) implements cost-cutting measures in order to continue working on the investigational device exemption (IDE) filing with the FDA
- In order to preserve capital for the strategic review process announced last week, workers will be temporarily laid off so the company can continue with the IDE filing at the FDA
- The US subsidiary will be temporarily furloughing approximately 40 employees at its Chapel Hill, North Carolina facility effective immediately
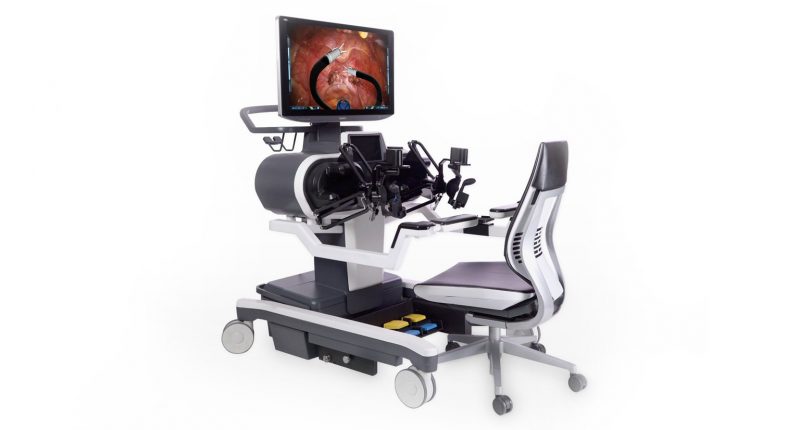
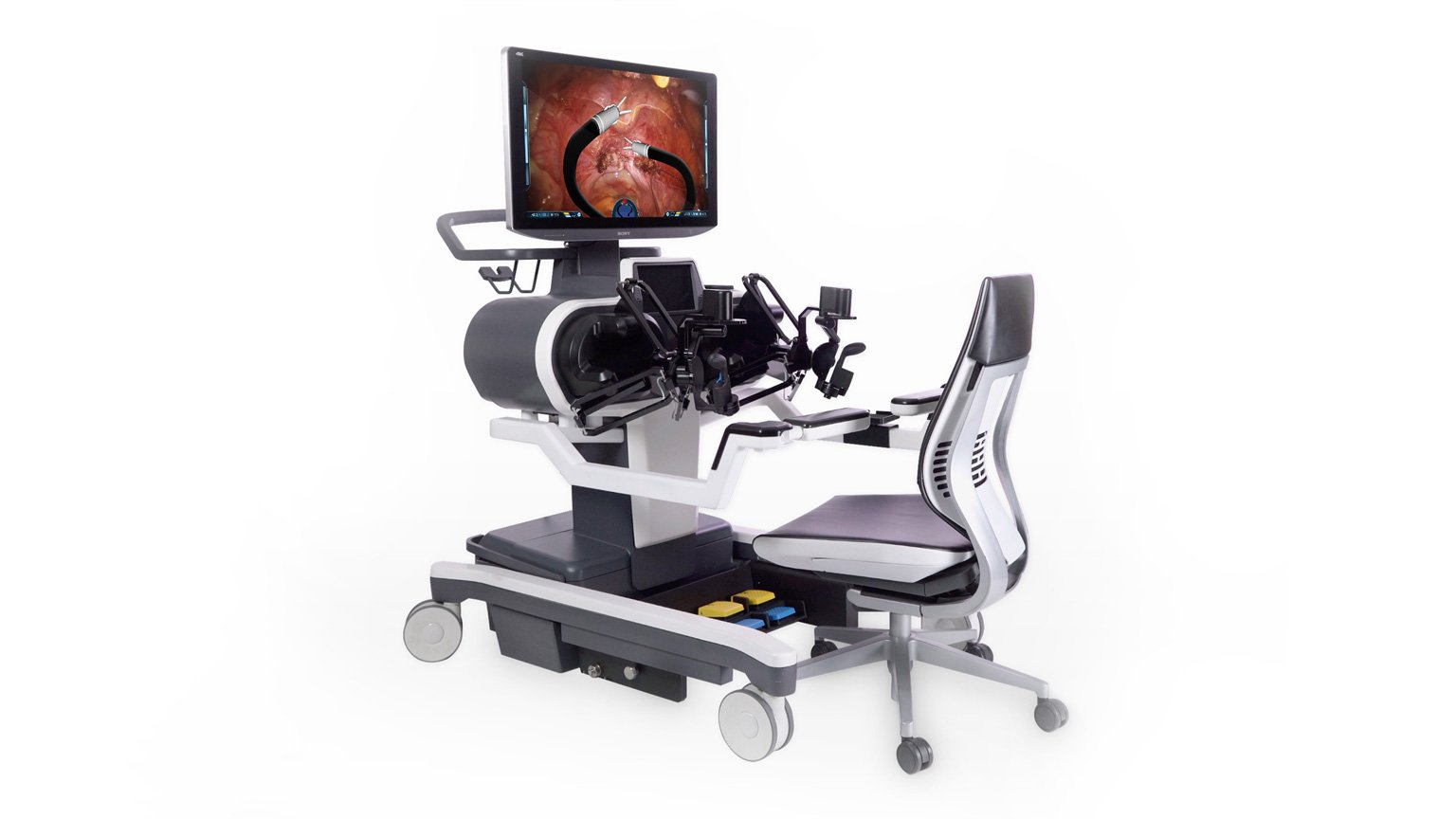
- Titan Medical (TMD) is a medical device company headquartered in Toronto with operations in North Carolina, focused on enhancing robotic-assisted surgery using innovative technology through a single access point
- Titan Medical (TMD) is trading at C$0.83 as of 3:45 pm ET
Titan Medical (TMD) implemented cost-cutting measures in order to continue working on the investigational device exemption (IDE) filing with the FDA.
In order to preserve capital for the IDE filing at the FDA announced last week, workers will be temporarily laid off. The company’s U.S. subsidiary will be temporarily furloughing approximately 40 employees at its Chapel Hill, North Carolina facility, effective immediately.
Cary Vance, Titan’s President and CEO, commented on the news.
“While the company works through the strategic review process, and in view of the company’s capital position and the challenging market conditions, we have had to make the extremely difficult decision to place some of our employees on furlough. These employees have made valuable contributions to the development of robotic-assisted technologies. As we have communicated to the impacted employees, we look forward to bringing our employees back to work if and as soon as possible. While this is not the news we would want to have been sharing, it is a necessary measure that allows us to strategically maintain a workforce that supports the strategic review process while continuing to make progress towards an IDE filing.”
The company will limit work to tasks related to the strategic review process, the IDE filing and fulfilling certain other contractual development and supply obligations. The cost-cutting measures will impact the timing and costs of the company’s milestones, and the company is therefore withdrawing all forward-looking statements included in its continuous disclosure documents with respect to the cost and timing of the development of its Enos surgical system.
Titan Medical (TMD) is a Canadian medical device company with operations in North Carolina focused on enhancing robotic-assisted surgery using innovative technology through a single access point.
Titan Medical (TMD) is trading at C$0.83 at 3:45 pm ET.