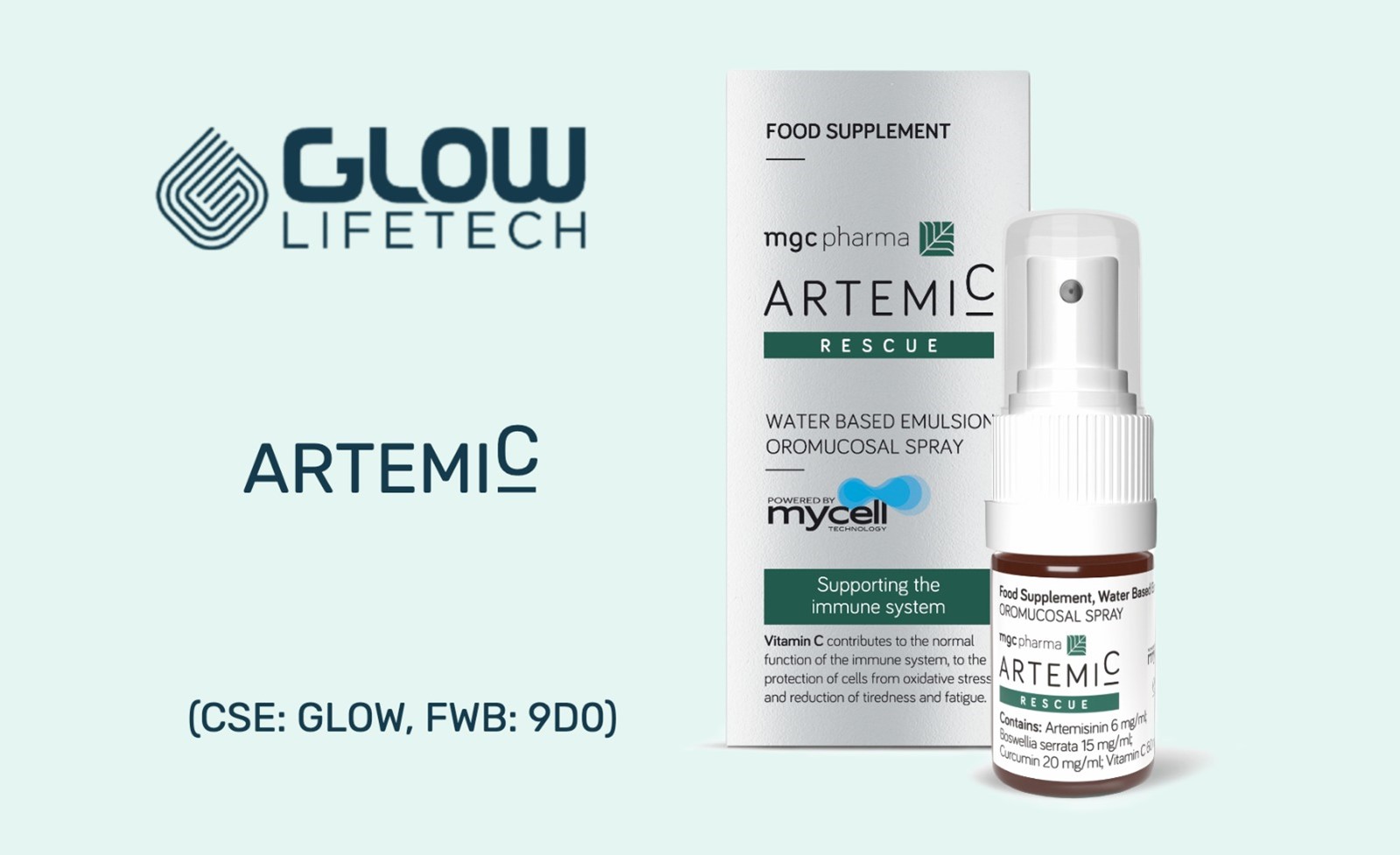
- Glow LifeTech submitted an application to Health Canada to obtain produce licenses for its Natural Health Product (NHP), ArtemiC™
- ArtemiC™ was submitted to Health Canada’s Natural and Non-prescription Health Products Directorate (NNHPD) on Mar 11, 2021
- With Health Canada approval and favourable clinical results, ArtemiC™ could become an all-natural solution to aid patient recovery
- Glow LifeTech is a Canadian-based biotechnology company focused on producing nutraceutical and cannabinoid-based products
- Glow LifeTech Corp. (GLOW) opened trading at C$0.54 per share
Relay Medical Corp (RELA), a shareholder of Glow LifeTech (GLOW) is pleased to provide an update on Glow LifeTech Corp.
On April 27, Glow submitted an application to Health Canada to obtain produce licenses for its Natural Health Product (NHP), ArtemiC™, which reported successful results from a COVID-19 Phase II clinical trial.
ArtemiC™ was submitted to Health Canada’s Natural and Non-prescription Health Products Directorate (NNHPD) on Mar 11, 2021. The application, which is currently under review by Health Canada, included ArtemiC™, which Glow LifeTech received exclusive rights to in early February.
“Canada has the potential to set the world stage for COVID-19 Natural Health Products. With Health Canada approval and favorable clinical results, ArtemiC™ could become an evidence backed all-natural solution to aid patient recovery,” stated Yoav Raiter, CEO, Relay Medical Corp.
“Glow has exclusive rights to market, sell, and distribute ArtemiC™ in Canada, the United States, Mexico, and all Caribbean countries as a food supplement (nutraceutical, dietary supplement, and natural health product).”
Glow LifeTech is a Canadian-based biotechnology company focused on producing nutraceutical and cannabinoid-based products.
Glow LifeTech Corp. (GLOW) opened trading at C$0.54 per share.