Treating heart disease and preventing coronary deaths have always been high priorities in pharmaceutical research. The rise of “Sudden Adult Death Syndrome” and a dramatic spike in inflammatory heart conditions among (otherwise) healthy young adults has added new urgency to this research.
Cardiol Therapeutics Inc. (NASDAQ:CRDL / TSX:CRDL) is a NASDAQ-listed life sciences company advancing its proprietary cannabidiol-based research to develop anti-inflammatory (and anti-fibrotic) therapies for the treatment of heart diseases.
On November 7, 2022, the Company announced study results demonstrating:
…that pharmaceutically manufactured cannabidiol (the active pharmaceutical ingredient in CardiolRx™) significantly reduces pericardial effusion and thickening in a pre-clinical model of acute pericarditis and significantly suppresses the secretion of key inflammatory markers interleukin-1β (“IL-1β”) and interleukin-6 (“IL-6”) in vitro.
CardiolRx™ is an oral cannabinoid formulation, manufactured to cGMP standards. Cardiol Therapeutics has already successfully completed a Phase I clinical study on CardiolRx™. The Company has also expressed its intentions to seek Orphan Drug status for CardiolRx™.
Pericarditis and myocarditis are the two categories of inflammatory heart disease that are currently attracting the most medical attention due to dramatically elevated rates of occurrence – especially in younger males.

“Pericardial effusion” is a build-up of fluid in the space around the heart, generally due to heart inflammation.
The significant reduction in the build-up of this fluid and news that CardiolRx™ “significantly suppresses…key inflammatory markers” are strong indicators of the potential efficacy of this drug therapy.
Study results were presented by Cardiol Therapeutics’ R&D partners from Virginia Commonwealth University (VCU). The setting was The American Heart Association Scientific Sessions 2022 (AHA2022).
Dr. Andrew Hamer, Chief Medical Officer and Head of Research and Development for Cardiol Therapeutics, framed the study results for investors.
“The data presented at the AHA2022 by our research collaborators from VCU provides additional rationale for our recent decision to prioritize our Phase II pilot study in patients with recurrent pericarditis and offers new insight into the molecular targets and mechanism of action of CardiolRx™.”
The study’s authors also observed that cannabidiol was shown in vitro to “significantly inhibit the transcription of IL-1β and NLRP3, as measured by mRNA expression”.
Cardiol Therapeutics is targeting the NLRP3 “inflammasome” with its R&D as this is a “large multiprotein complex that regulates inflammatory responses of the innate immune system”.
Dr. Hamer explained the significance of these inflammasomes as they relate to pericarditis.
“Pericarditis results from inflammation of the pericardium, the sac that surrounds the heart, and is manifested clinically by chest pain and by imaging signs of pericardial effusion and thickening. Significant accumulation of pericardial fluid and scarring can progress to life-threatening constriction of the heart. It is recognized that pericarditis is associated with aberrant inflammasome activation.”
Cardiol Therapeutics has filed new patent applications with the US patent office in connection with these new findings.
The Company has already received IND approval from the FDA to conduct two Phase II clinical studies to evaluate the safety and efficacy of CardiolRx™.
One of these Phase II studies will be a multi-national, double-blind trial of acute myocarditis patients using CardiolRx™ (with half the sample population being administered a placebo). This is the Company’s “ARCHER trial”.
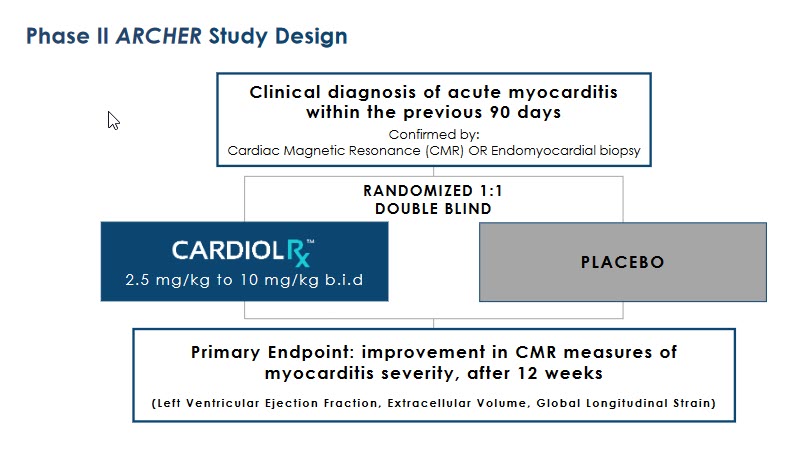
The primary endpoint will be an improvement in patients’ myocarditis, defined as improved Cardiac Magnetic Resonance (CMR) measurements of myocarditis severity. The duration of the study is 12 weeks.
The second Phase II multi-center open-label pilot study in recurrent pericarditis that will involve 25 patients from several major clinical centers in the United States. Recurrent pericarditis is associated with a number of symptoms, including debilitating chest pain, fatigue and shortness of breath.
Recurrent pericarditis results in physical limitations that lead to reduced quality of life and increased emergency department visits and hospitalizations. Existing treatment options are both very limited and extremely costly.
The primary endpoint for this second Phase II study will be based on in-patient reporting of pericarditis pain, using an 11-point numeric rating scale. That evaluation will be conducted after 8 weeks.
A secondary endpoint for the study will be to make a similar evaluation after 26 weeks. The evaluation will include measuring “circulating levels of C-reactive protein”. This is a commonly used marker of inflammation.
Complimenting this research, the Company has also announced that it is developing a novel subcutaneously administered drug formulation of cannabidiol, aimed as a treatment for heart failure. This is an even larger treatment market, with associated healthcare costs in the United States alone that exceed $30 billion per year.
As healthcare systems turn their attention toward treating non-Covid related health conditions, the treatment of pericarditis and myocarditis (and preventing heart failure) is sure to garner increasing attention.
As these treatment markets swell in size, that will, in turn, draw the interest of increasing numbers of investors.
Cardiol Therapeutics offers investors a very attractive share structure to go along with its NASDAQ listing. With the recent carnage in markets dragging the Company’s market cap down below CAD$50 million, Cardiol Therapeutics currently provides investors with a very attractive entry point at present.
As governments and healthcare systems move on from the Covid-19 pandemic to new challenges in healthcare, that’s the cue for investors to start looking for new opportunities in pharma.
FULL DISCLOSURE: This is a paid article by The Market Herald.





